Chapter 20: Q56P (page 1038)
Show how you would synthesize from each compound. You may use any necessary reagents.
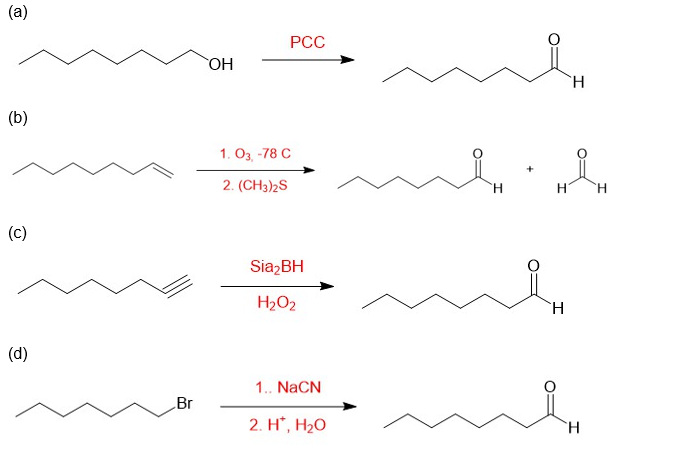
(a)
(b)
(c)
(d)
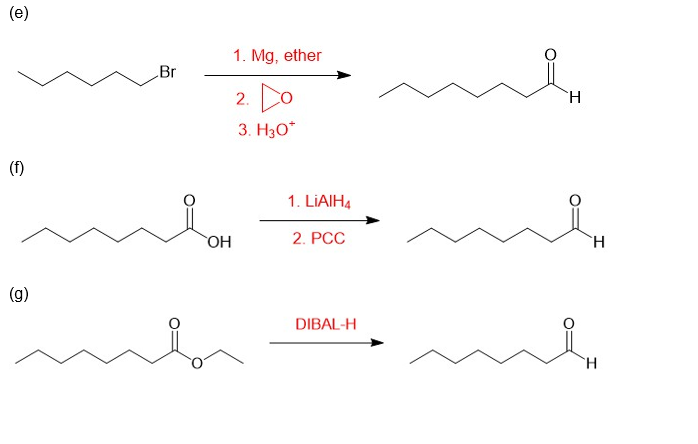
(e)
(f)
(g)
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q56P (page 1038)
Show how you would synthesize from each compound. You may use any necessary reagents.
(a)
(b)
(c)
(d)
(e)
(f)
(g)


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
Predict the compound in each pair that will undergo solvolysis (in aqueous ethanol) more rapidly.
(a)
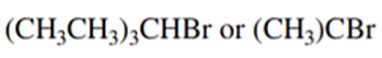
(b)
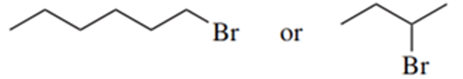
(c)
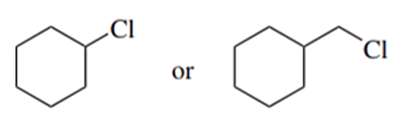
(d)
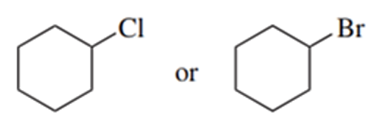
(e)
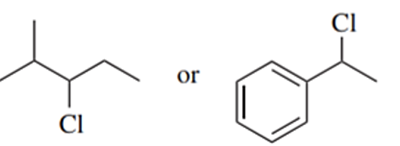
(f)
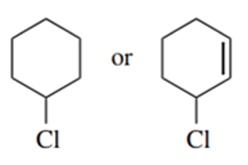
Tributylin hydride (Bu3SnH)is used synthetically to reduce alkyl halides, replacing a halogen atom with hydrogen. Free-radical initiators promote this reaction, and free-radical inhibitors are known to slow or stop it. You job is to develop a mechanism, using the following reaction as an example.
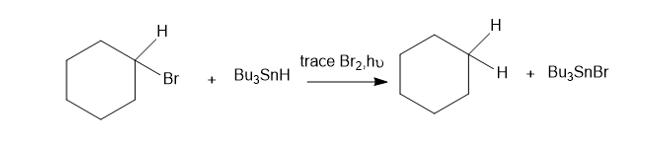
The following bond-dissociation enthalpies may be helpful:
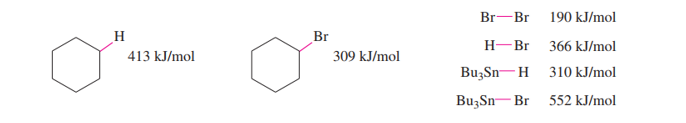
(a) Propose initiation and propagation steps to account for this reaction.
(b) Calculate values of Δ�� for your proposed steps to show that they are energetically feasible. (Hint: A trace of Br2 and light suggests it’s there only as an initiator, to create Brradicals. Then decide which atom can be extracted most favorably from the starting materials by the Brradical. That should complete the initiation. Finally, decide what energetically favored propagation steps will accomplish the reaction).
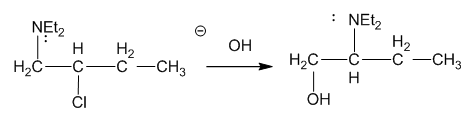
The following reaction takes place under second-order conditions (strong nucleophile), yet the structure of the product shows rearrangement. Also, the rate of this reaction is several thousand times faster than the rate of substitution of hydroxide ion on 2-chlorobutane under similar conditions. Propose a mechanism to explain the enhanced rate and rearrangement observed in this unusual reaction. (“Et” is the abbreviation for ethyl.
(a) How many asymmetric carbon atoms are there in an aldotetrose? Draw all the aldotetrose stereoisomers.
(b) How many asymmetric carbon atoms are there in a ketotetrose? Draw all the ketotetrose stereoisomers.
(c) How many asymmetric carbon atoms and stereoisomers are there for an aldohexose? For a ketohexose?
What do you think about this solution?
We value your feedback to improve our textbook solutions.