Chapter 20: Q30P (page 1038)
Rank the following radicals in decreasing order of stability. Classify each as primary, secondary, or tertiary.
(a)
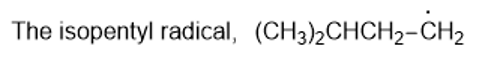
(b)

(c)
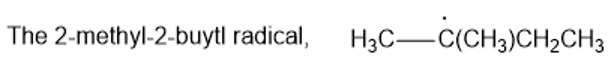
(d)
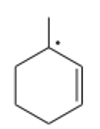
Short Answer
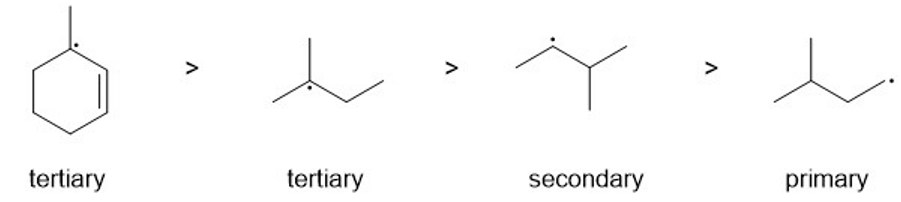
radicals in order from most stable to least stable
Learning Materials
Features
Discover
Chapter 20: Q30P (page 1038)
Rank the following radicals in decreasing order of stability. Classify each as primary, secondary, or tertiary.
(a)
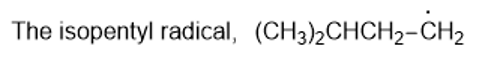
(b)

(c)
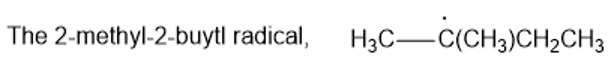
(d)


radicals in order from most stable to least stable
All the tools & learning materials you need for study success - in one app.
Get started for free
Dimethylamine(CH3) 2 NHhas a molecular weight of 45 and a boiling point of 7.40C. Trimethylamine, (CH3) 3N, has a higher molecular weight (59) but a lower boiling point(3.50C) . Explain this apparent discrepancy.
Question:
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
The reaction of tert-butyl alcohol with concentrated HCl goes by the SN1 mechanism. Write a mechanism for this reaction.
Grignard reagents add to carbonate esters as they add to other esters.
(a) Predict the major product of the following reaction.
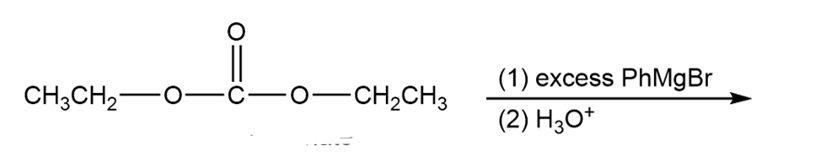
(b) Show how you would synthesize 3-ethylpentan-3-ol using diethyl carbonate and ethyl bromide as your only organic reagents.
(c) Diethyl carbonate is a liquid reagent that is easy to handle. In contrast, phosgene is a highly toxic and corrosive gas. Show how you might use diethyl carbonate instead of phosgene to make Lexan®. Also, show how you might use diethyl carbonate instead of methyl isocyanate to make Sevin® insecticide.
What do you think about this solution?
We value your feedback to improve our textbook solutions.