Chapter 6: Q36P (page 329)
Give two syntheses for (CH3)2CH-OCH3, and explain which synthesis is better.
Short Answer
Two methods for syntheses of (CH3)2CH-OCH3,
- Williamson synthesis
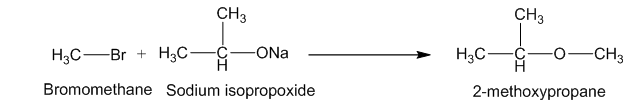
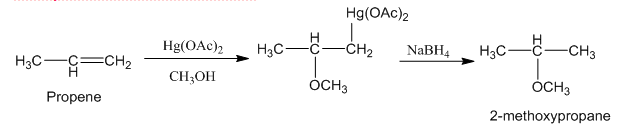
2. Alkoxymercuration-Demercuration
Learning Materials
Features
Discover
Chapter 6: Q36P (page 329)
Give two syntheses for (CH3)2CH-OCH3, and explain which synthesis is better.
Two methods for syntheses of (CH3)2CH-OCH3,

2. Alkoxymercuration-Demercuration

All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the compound in each pair that will undergo the SN2 reaction faster.
(a.)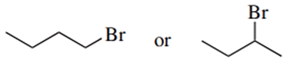
(b.)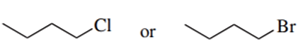
(c.)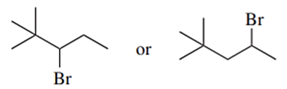
(d.)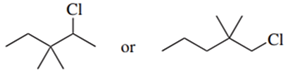
(e.)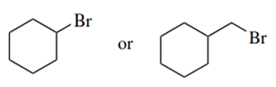
(f.)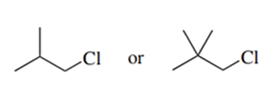
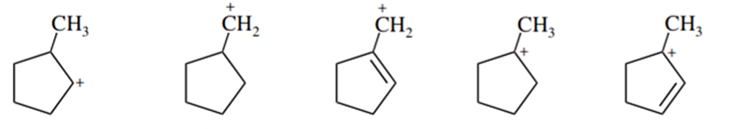
List the following carbocations in decreasing order of their stability.
When water is shaken with hexane, the two liquids separate into two phases. Which compound is present in the top phase, and which is present in the bottom phase? When water is shaken with chloroform, a similar two-phase system results. Again, which compound is present in each phase? Explain the difference in the two experiments. What do you expect to happen when water is shaken with ethanol (CH3CH2OH)?
Strawberry growers have used large quantities of methyl bromide (b.p. 4°C) to sterilize the soil before planting their crops. Like some of the freons, methyl bromide can diffuse up into the stratosphere, where it damages the protective ozone layer. Agricultural chemists have suggested using methyl iodide (b.p. 43°C) as a replacement for methyl bromide. Why is methyl iodide likely to be more toxic to agricultural pests (and people) than methyl bromide? Why is methyl iodide less likely to reach the stratosphere than methyl bromide?
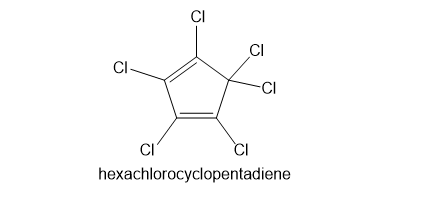
Kepone, aldrin, and chlordane are synthesized from hexachlorocyclopentadiene and other five-membered ring compounds. Show how these three pesticides are composed of two five-membered rings.
What do you think about this solution?
We value your feedback to improve our textbook solutions.