Chapter 20: Q20.74P (page 919)
For the reaction , calculate Kp atof.
Short Answer
The Kp value is
Learning Materials
Features
Discover
Chapter 20: Q20.74P (page 919)
For the reaction , calculate Kp atof.
The Kp value is
All the tools & learning materials you need for study success - in one app.
Get started for free
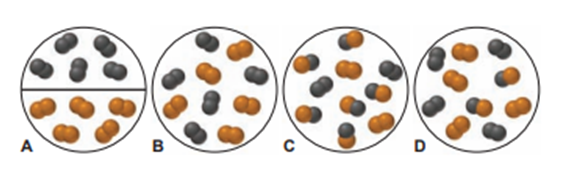
The scenes and the graph relate to the reaction of(black) with (orange) to form .
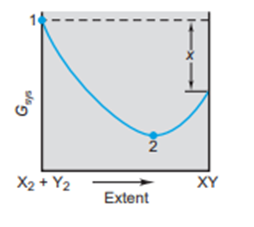
(a) If reactants and products are in their standard states, what quantity is represented on the graph by x?
(b) Which scene represents point 1? Explain.
(c) Which scene represents point 2? Explain.
Sulfur dioxide is released in the combustion of coal. Scrubbers use aqueous slurries of calcium hydroxide to remove the from flue gases. Write a balanced equation for this reaction and calculate role="math" localid="1663363200253" at 298 K [ of J/mol K].
According to advertisements, "a diamond is forever."
(a) Calculate , and at 298k for the phase change Diamond graphite.
(b) Given the conditions under which diamond jewelry is normally kept, argue for and against the statement in the ad.
(c) Given the answers in part (a), what would need to be done to make synthetic diamonds from graphite?
(d) Assumingrole="math" localid="1663313565397" androle="math" localid="1663313547943" do not change with temperature, can graphite be converted to diamond spontaneously at 1 atm?
How can be relatively independent of ifof each reactant and product increases with ?
Consider the oxidation of carbon monoxide:
(a) Predict the signs of and . Explain.
(b) Calculate by two different methods
What do you think about this solution?
We value your feedback to improve our textbook solutions.