Chapter 20: Q20.69 P (page 919)
Calculate K at for each reaction:
(a) data-custom-editor="chemistry"
(b)
(c)
Short Answer
(a) The value is .
(b) The value is .
(c) The value is .
Learning Materials
Features
Discover
Chapter 20: Q20.69 P (page 919)
Calculate K at for each reaction:
(a) data-custom-editor="chemistry"
(b)
(c)
(a) The value is .
(b) The value is .
(c) The value is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Without consulting Appendix B, arrange each group in order of decreasing standard molar entropy (S°). Explain.
(a) Mg metal, Ca metal, Ba metal
(b) Hexane , benzene , cyclohexane
(c)(g), (g), (g).
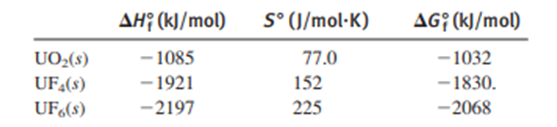
To prepare nuclear fuel, U3O8 ("yellow cake") is converted to UO2(NO3)2, which is then converted to UO3 and finally UO2.The fuel is enriched (the proportion of the is increased) by a two-step conversion of UO2 into UF6, a volatile solid, followed by a gaseous-diffusion separation of the isotopes:
Calculate for the overall process at .
When heated, the DNA double helix separates into two random-coil single strands. When cooled, the random coils reform the double helix: double helix 2 random coils.
(a) What is the sign offor the forward process? Why?
(b) Energy must be added to overcome H bonds and dispersion forces between the strands. What is the sign offor the forward process whenis smaller thanrole="math" localid="1663303287852" ?
(c) Write an expression that shows T in terms ofandwhen the reaction is at equilibrium. (This temperature is called the melting temperature of the nucleic acid.)
Use Appendix B to determine theof
a) Write a balanced equation for the gaseous reaction between N2O5 and F2 to form NF3 and O2.
b) Determine
c) Find role="math" localid="1663390048995" if
What do you think about this solution?
We value your feedback to improve our textbook solutions.