Chapter 20: Q20.58 P (page 918)
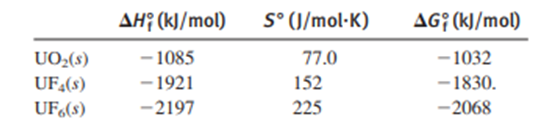
One reaction used to produce small quantities of pure is
(a) Determine and for the reaction at .
(b) Assuming that these values are relatively independent of temperature, calculate at , and.
(c) What is the significance of the different values of ?
Short Answer
- The enthalpy value is and entropy value is .
The enthalpy and entropy changes values are a positive sign for indicates the formation of methanol is favored at a given temperature.
- The standard free energy values are and .
- Given the reaction the enthalpy and entropy values are positive at high temperatures.