Chapter 20: Q20.51 P (page 880)
Calculate for each reaction using values:
(a)
(b)
(c)
Short Answer
- The Gibbs free energy value is .
- The Gibbs free energy value is .
- The Gibbs free energy value is.
Learning Materials
Features
Discover
Chapter 20: Q20.51 P (page 880)
Calculate for each reaction using values:
(a)
(b)
(c)
All the tools & learning materials you need for study success - in one app.
Get started for free
When heated, the DNA double helix separates into two random-coil single strands. When cooled, the random coils reform the double helix: double helix 2 random coils.
(a) What is the sign offor the forward process? Why?
(b) Energy must be added to overcome H bonds and dispersion forces between the strands. What is the sign offor the forward process whenis smaller thanrole="math" localid="1663303287852" ?
(c) Write an expression that shows T in terms ofandwhen the reaction is at equilibrium. (This temperature is called the melting temperature of the nucleic acid.)
For the gaseous reaction of carbon monoxide and chlorine to form phosgene :
(a) Calculate at and .
(b) Assuming that and change little with temperature, calculate at .
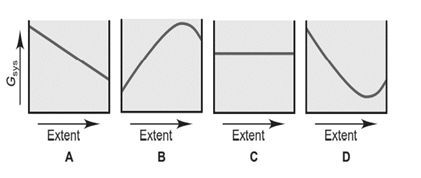
A chemical reaction, such as HI forming from its elements, can reach equilibrium at many temperatures. In contrast, a phase change, such as ice melting, is in equilibrium at a given pressure only at the melting point. Each of the graphs below depicts vs. extent of change.
(a) Which graph depicts how changes for the formation of HI? Explain.
(b) Which graph depicts how changes as ice melts at and 1atm? Explain.
What is the advantage of calculating free energy changes rather than entropy changes to determine reaction spontaneity?
Among the many complex ions of cobalt are the following:
where "en" stands for ethylenediamine, . Six bonds are broken and six bonds are formed in this reaction, so localid="1663319605453" ; yet . What are the signs of localid="1663319705259" and ? What drives the reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.