Chapter 20: Q20.30 P (page 917)
For the reaction depicted in the molecular scenes, X is red and Y is green.
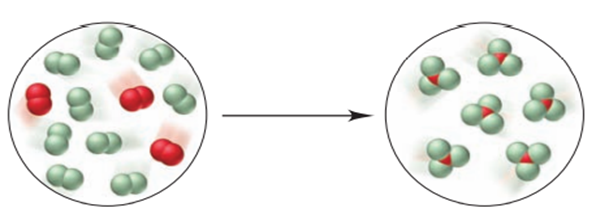
- Write a balanced equation.
- Determine the sign of Δł§.
- Which species has the highest molar entropy?
Short Answer
- The balanced equation is: .
- The sign is obtained as: .
- The species that has the highest molar entropy is: .

