Chapter 11: Q11.57CP (page 435)
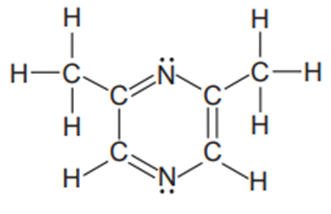
The compound 2,6-dimethyl pyrazine (below) gives chocolate its odor and is used in flavourings.
- Which atomic orbitals mix to form the hybrid orbitals ?
- In what type of hybrid orbital do the lone pairs reside?
- Is in hybridized the same as any in the ring? Explain.
Short Answer
Hybridization provides a hybrid state for the overlapping of electrons to maximize stability.
The answer for
a)oneand two role="math" localid="1658940566747" atomic orbitals.
b) orbital
c) No