Chapter 11: Q11.41CP (page 434)
Use partial orbital diagrams to show how the atomic orbitals of the central atom lead to the hybrid orbitals in:
Learning Materials
Features
Discover
Chapter 11: Q11.41CP (page 434)
Use partial orbital diagrams to show how the atomic orbitals of the central atom lead to the hybrid orbitals in:
All the tools & learning materials you need for study success - in one app.
Get started for free
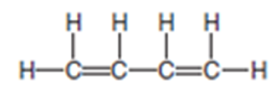
Butadiene is a colourless gas used to make synthetic rubber and many other compounds.
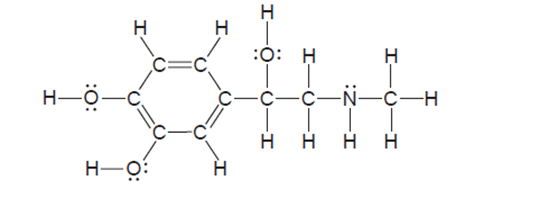
Epinephrine (or adrenaline; see structure) is a naturally occurring hormone that is also manufactured commercially for use as a heart stimulant, a nasal decongestant, and to treat glaucoma.
2-Butene () is a starting material in the manufacture of lubricating oils and many other compounds. Draw two different structures for 2-butene, indicating the sigma and pi bonds in each.
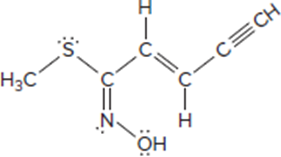
An organic chemist synthesizes the molecule below:

Some species with two oxygen atoms only are the oxygen molecule , the peroxide ion, , the superoxide ion, , and the dioxygenyl ion, Draw an MO diagram for each, rank them in order of increasing bond length, and find the number of unpaired electrons in each.
What do you think about this solution?
We value your feedback to improve our textbook solutions.