Chapter 10: Q10.78CP (page 408)
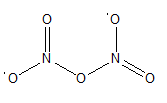
An oxide of nitrogen is N by mass, has a molar mass of 108 g/mol, and contains no nitrogen-nitrogen or oxygen- oxygen bonds. Draw its Lewis structure, and name it.
Short Answer
Its Lewis structure is,
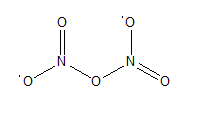
The name of the compound is dinitrogen pentoxide.