When phosphate is protonated to generate hydrogen phosphate () and dihydrogen phosphate (), acid rain increases the leaching of phosphates, PO, into groundwater ().
a)is a soluble terrestrial phosphate rock, and the solubility of salt may be calculated using the appropriate solubility constant.
has a solubility product constant ofin pure water (pH) according to Appendix C, whereas the solubility product constant may be represented as:
As calcium phosphate is a solid, its concentration is one, hence it may be left out of the equation:
If we assume that the starting concentration of was one and that the ions concentration was , then after the reaction, some concentration was acquired by ions and some concentration was obtained by ions at equilibrium, as shown in Table below:
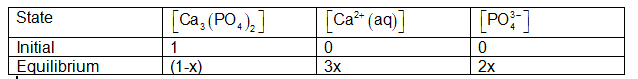
As a result, the equilibriummay be expressed as
Then solving the x equation as:
localid="1663309379461"
Therefore, the calcium phosphate has a solubility of M at pH .


