Chapter 22: Q22.82CP (page 1021)
Like heavy water (), so-called “semi-heavy water” (HDO) undergoes H/D exchange. The scenes below depict an initial mixture of HDO and reaching equilibrium.
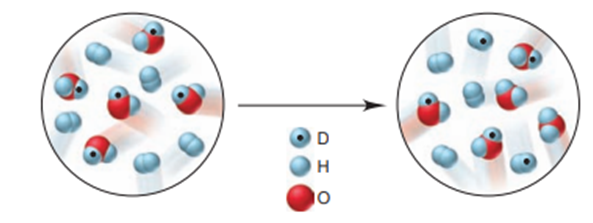
a) Write the balanced equation for the reaction. (b) Is the value of K greater or less than? (c) If each molecule depicted represents0.10M, calculate K.
Short Answer
a) The balanced equation for the reaction is: .
b) The value of K is: .
c) The value of K is obtained as: .

