Chapter 2: Q96P (page 40)
Assuming acid strength relates directly to a number of atoms bonded to the central atom, rank role="math" localid="1663308780600" ; ; and in order of decreasing acid strength.
Short Answer
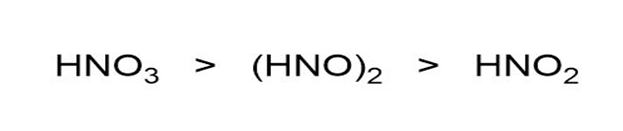
Learning Materials
Features
Discover
Chapter 2: Q96P (page 40)
Assuming acid strength relates directly to a number of atoms bonded to the central atom, rank role="math" localid="1663308780600" ; ; and in order of decreasing acid strength.
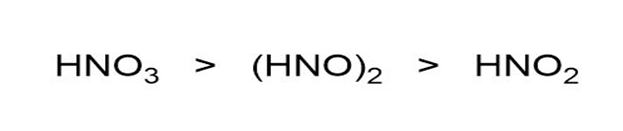
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify the mass law that each of the following observations demonstrates, and explain your reasoning:
(a) A sample of potassium chloride from Chile contains the same percent by mass of potassium as one from Poland.
(b) A flashbulb contains magnesium and oxygen before use and magnesium oxide afterward, but its mass does not change.
(c) Arsenic and oxygen form one compound that is 65.2 mass % arsenic and another that is 75.8 mass % arsenic.
State the mass law(s) demonstrated by the following experimental results, and explain your reasoning:
Experiment 1: A student heats 1.00 g of a blue compound and obtains 0.64 g of a white compound and 0.36 g of a colour less gas.
Experiment 2: A second student heats 3.25 g of the same blue compound and obtains 2.08 g of a white compound and 1.17 g of a colour less gas.
Give the name and formula of the compound formed from the following elements
a.
b.
c.
The tap water found in many areas of the United States leaves white deposits when it evaporates. Is this tap water a mixture or a compound? Explain.
Define atomic number and mass number which can vary without changing the identity of the element?
What do you think about this solution?
We value your feedback to improve our textbook solutions.