Chapter 2: Q83P (page 84)
For what type of compound are we unable to write a molecular formula?
Short Answer
Ionic compounds are not represented by molecular formula but are represented by empirical formula.
Learning Materials
Features
Discover
Chapter 2: Q83P (page 84)
For what type of compound are we unable to write a molecular formula?
Ionic compounds are not represented by molecular formula but are represented by empirical formula.
All the tools & learning materials you need for study success - in one app.
Get started for free
List two differences between a compound and a mixture.
Fluorite, a mineral of calcium, is a compound of the metal with fluorine. Analysis shows that a 2.76-g sample of fluorite contains 1.42 g of calcium. Calculate the (a) mass of fluorine in the sample; (b) mass fractions of calcium and fluorine in fluorite; (c) mass percents of calcium and fluorine in fluorite.
How can ionic compounds be neutral if they consist of positive and negative ions?
Each scene below represents a mixture. Describe each one in terms of the number of elements and/or compounds present.


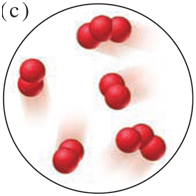
Classify each substance in Problem 2.3 as an element, compound, or mixture, and explain your answers.
What do you think about this solution?
We value your feedback to improve our textbook solutions.