Chapter 2: Q59 P (page 40)
Give systematic names for the following formulas:
Short Answer
a)- potassium di cyano argentate(l)
b)- sodium tetra chlorocadmate (II)
c)- tetra ammine aqua bromo cobalt (III)
Learning Materials
Features
Discover
Chapter 2: Q59 P (page 40)
Give systematic names for the following formulas:
a)- potassium di cyano argentate(l)
b)- sodium tetra chlorocadmate (II)
c)- tetra ammine aqua bromo cobalt (III)
All the tools & learning materials you need for study success - in one app.
Get started for free
The tap water found in many areas of the United States leaves white deposits when it evaporates. Is this tap water a mixture or a compound? Explain.
Each scene below represents a mixture. Describe each one in terms of the number of elements and/or compounds present.


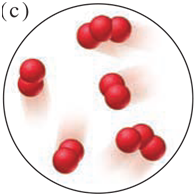
State the mass law(s) demonstrated by the following experimental results, and explain your reasoning:
Experiment 1: A student heats 1.27 g of copper and 3.50 g of iodine to produce 3.81 g of a white compound; 0.96 g of iodine remains.
Experiment 2: A second student heats 2.55 g of copper and 3.50 g of iodine to form 5.25 g of a white compound, and 0.80 g of copper remains.
Choose the correct answer. The difference between the mass number of an isotope and its atomic number is (a)directly related to the identity of the element; (b) the number of electrons; (c) the number of neutrons; (d) the number of isotopes.
Explain the following statement: The smallest particles unique to a compound cannot be atoms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.