Chapter 2: Q3.115CP (page 40)
Hydrogen-containing fuels have a “fuel value” based on their mass % H. Rank the following compounds from highest mass % H to lowest: ethane, propane, benzene, ethanol, cetyl palmitate (whale oil, C32H64O2).
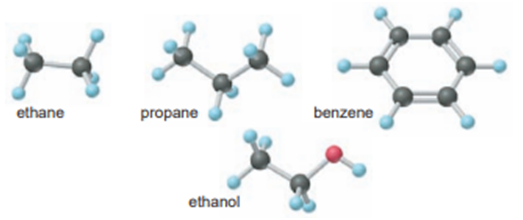
Short Answer
Thecompound from the highest mass percent to the lowest is:


