Chapter 2: Q24.75P (page 40)
AH nucleus decays with an energy ofMeV. Convert this energy into (a) electron volts; (b) joules.
Learning Materials
Features
Discover
Chapter 2: Q24.75P (page 40)
AH nucleus decays with an energy ofMeV. Convert this energy into (a) electron volts; (b) joules.
All the tools & learning materials you need for study success - in one app.
Get started for free
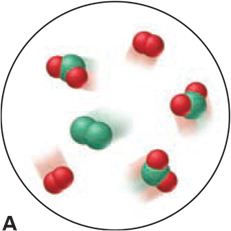
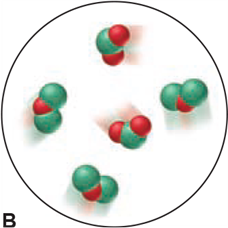
Which of the following scenes illustrate(s) the fact that compounds of chlorine (green) and oxygen (red) exhibit the law of multiple proportions? Name the compounds.

The elements inGroupsandare all quite reactive. What is the major difference between them?
Can the relative amounts of the components of a mixture vary? Can the relative amounts of the components of a compound vary? Explain.
Choose the correct answer. The difference between the mass number of an isotope and its atomic number is (a)directly related to the identity of the element; (b) the number of electrons; (c) the number of neutrons; (d) the number of isotopes.
Explain the following statement: The smallest particles unique to an element may be atoms or molecules.
What do you think about this solution?
We value your feedback to improve our textbook solutions.