Chapter 2: Q15.54 P (page 40)
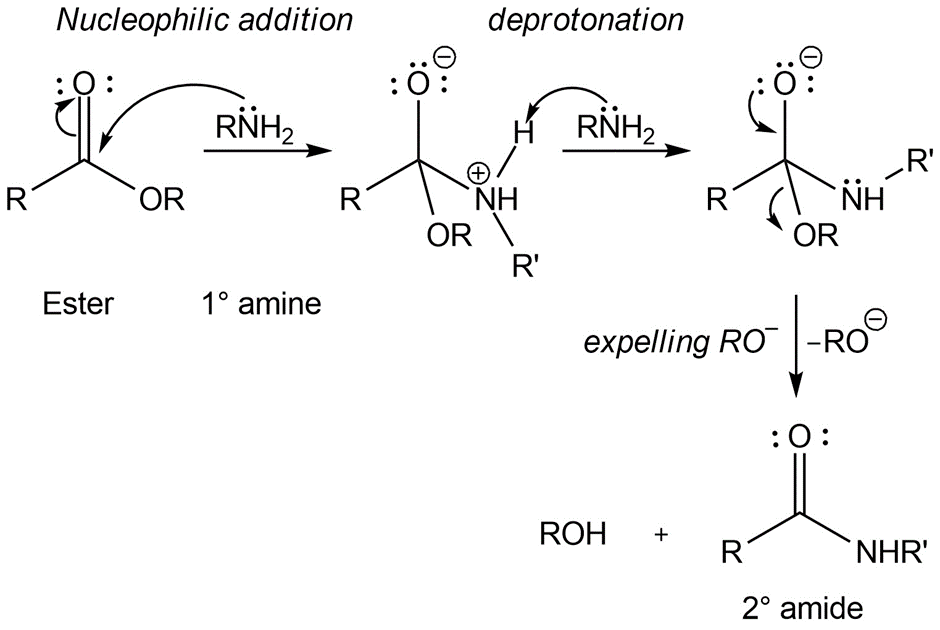
Many substitution reactions involve an initial electrostatic attraction between reactants. Show where this attraction arises in the formation of an amide from an amine and an ester
Short Answer
Formation of amide:
Learning Materials
Features
Discover
Chapter 2: Q15.54 P (page 40)
Many substitution reactions involve an initial electrostatic attraction between reactants. Show where this attraction arises in the formation of an amide from an amine and an ester
Formation of amide:

All the tools & learning materials you need for study success - in one app.
Get started for free
What class of elements lies along the “staircase” line in the periodic table? How do their properties compare with those of metals and nonmetals?
The radii of the lithium and magnesium ions are 76 pm and 72 pm, respectively. Which compound has stronger ionic attractions, lithium oxide or magnesium oxide?
The radii of the sodium and potassium ions are 102 pm and 138 pm, respectively. Which compound has stronger ionic attractions, sodium chloride or potassium chloride?
Chlorine has two naturally occurring isotopes, 35Cl and 37Cl. What is the mass number of each isotope? How many protons, neutrons, and electrons are present in each?
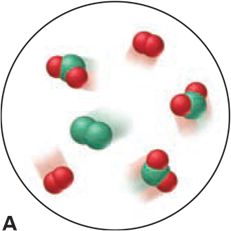
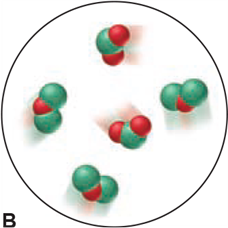
Which of the following scenes illustrate(s) the fact that compounds of chlorine (green) and oxygen (red) exhibit the law of multiple proportions? Name the compounds.

What do you think about this solution?
We value your feedback to improve our textbook solutions.