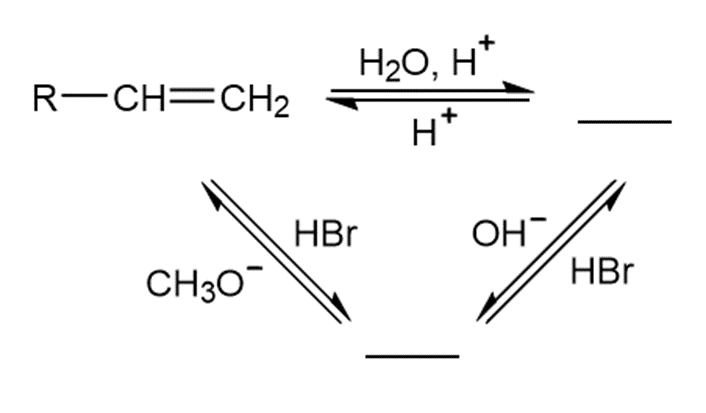
Chapter 2: Q15.51 P (page 40)
Fill in each blank with a general formula for the type of compound formed:
Short Answer
Reactions followed as follows:
Learning Materials
Features
Discover
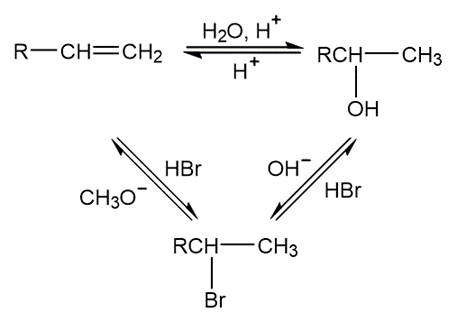
Chapter 2: Q15.51 P (page 40)
Fill in each blank with a general formula for the type of compound formed:

Reactions followed as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
Zinc sulfide (ZnS) occurs in the zinc blend crystal structure.How many kilograms of Zn are in 3.82 kg of ZnS?
Show, with calculations, how the following data illustrate the law of multiple proportions:Compound 1: 77.6 mass % xenon and 22.4 mass % fluorineCompound 2: 63.3 mass % xenon and 36.7 mass % fluorine
Show, with calculations, how the following data illustrate thelaw of multiple proportions:Compound 1: 47.5 mass % sulfur and 52.5 mass % chlorineCompound 2: 31.1 mass % sulfur and 68.9 mass % chlorine
The following charges on individual oil droplets were obtained during an experiment similar to Millikan's. Determine a charge for the electron( in coulombs), and explain your answer:
; ; ; .
Choose the correct answer. The difference between the mass number of an isotope and its atomic number is (a)directly related to the identity of the element; (b) the number of electrons; (c) the number of neutrons; (d) the number of isotopes.
What do you think about this solution?
We value your feedback to improve our textbook solutions.