Chapter 2: Q152CP (page 88)
TNT (trinitrotoluene; below) is used as an explosive in construction. Calculate the mass of each element in 1.00 lb of TNT.
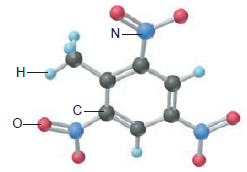
Short Answer
Themass of each element in 1 lb of TNT can be found, as:
Learning Materials
Features
Discover
Chapter 2: Q152CP (page 88)
TNT (trinitrotoluene; below) is used as an explosive in construction. Calculate the mass of each element in 1.00 lb of TNT.

Themass of each element in 1 lb of TNT can be found, as:
All the tools & learning materials you need for study success - in one app.
Get started for free
Classify each substance in Problem 2.3 as an element, compound, or mixture, and explain your answers.
Fill in the blanks:
(a) The symbol and atomic number of the heaviest alkaline earthmetal are ________ and ________.
(b) The symbol and atomic number of the lightest metalloidin Group 4A(14)are ________ and ________.
(c) Group 1B(11) consists of the coinage metals.The symbol and atomic mass of the coinage metal whose atoms have the fewest electrons are________ and ________.
(d) The symbol and atomic mass of the halogen in Period 4 are ________ and ________.
The following charges on individual oil droplets were obtained during an experiment similar to Millikan's. Determine a charge for the electron( in coulombs), and explain your answer:
For what type(s) of compound do we use Greek numerical prefixes in the name?
When Rutherford's coworkers bombarded gold foil with particles, they obtained results that overturned the existing (Thomson) model of the atom. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.