Chapter 2: Q113P (page 85)
Give the name, empirical formula and molecular mass of the molecule depicted in the figure.
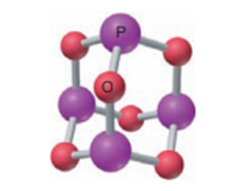
Short Answer
Molecular Name – Tetraphosphrous hexaoxide
Empirical formula -
Molecular Mass -
Learning Materials
Features
Discover
Chapter 2: Q113P (page 85)
Give the name, empirical formula and molecular mass of the molecule depicted in the figure.

Molecular Name – Tetraphosphrous hexaoxide
Empirical formula -
Molecular Mass -
All the tools & learning materials you need for study success - in one app.
Get started for free
A compound of copper and sulfur contains 88.39 g of metal and 44.61 g of nonmetal. How many grams of copper are in5264 kg of the compound? How many grams of sulfur?
When Rutherford's coworkers bombarded gold foil with particles, they obtained results that overturned the existing (Thomson) model of the atom. Explain.
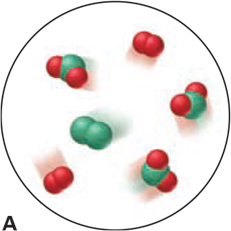
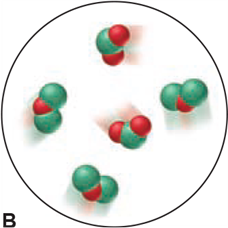
Which of the following scenes illustrate(s) the fact that compounds of chlorine (green) and oxygen (red) exhibit the law of multiple proportions? Name the compounds.

Dolomite is a carbonate of magnesium and calcium. Analysisshows that 7.81 g of dolomite contains 1.70 g of Ca. Calculate themass percent of Ca in dolomite. On the basis of the mass percentof Ca, and neglecting all other factors, which is the richer sourceof Ca, dolomite or fluorite (see Problem 2.20)?
When Rutherford's coworkers bombarded gold foil with particles, they obtained results that overturned the existing (Thomson) model of the atom. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.