Chapter 3: Q3.39P (page 132)
An oxide of nitrogen contains 30.45 mass %.
(a) What is the empirical formula of the oxide?
(b) If the molar mass iswhat is the molecular formula?
Short Answer
- The empirical formula is
- The molecular formula is
Learning Materials
Features
Discover
Chapter 3: Q3.39P (page 132)
An oxide of nitrogen contains 30.45 mass %.
(a) What is the empirical formula of the oxide?
(b) If the molar mass iswhat is the molecular formula?
All the tools & learning materials you need for study success - in one app.
Get started for free
Loss of atmospheric ozone has led to an ozone "hole" over Antartica. The process occurs in part by three consecutive reactions:
Chlorine monoxide forms ClOOCl
Manganese is a key component of extremely hard steel. The element occurs naturally in many oxides. A 542.3-g sample of a manganese oxide has an Mn/O ratio of 1.00/1.42 and consists of braunite (Mn2O3) and manganosite (MnO). (a) What masses of braunite and manganosite are in the ore? (b) What is the ratio Mn3+/Mn2+ in the ore?
Hydrocarbon mixtures are used as fuels. A252-g gaseous mixture of CH4 and C3H8 burns in excess O2, and 748 g of CO2 gas is collected. What is the mass % of CH4 in the mixture?
When powdered zinc is heated with sulfur, a violent reaction occurs, and zinc sulfide forms:
Some of the reactants also combine with oxygen in air to form zinc oxide and sulfur dioxide. When 83.2 g of Zn reacts with 52.4 g of S8, 104.4 g of ZnS forms. What is the percent yield of ZnS? (b) If all the remaining reactants combine with oxygen, how many grams of each of the two oxides form?
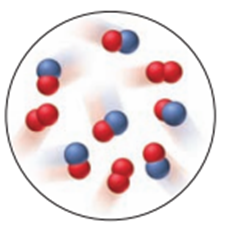
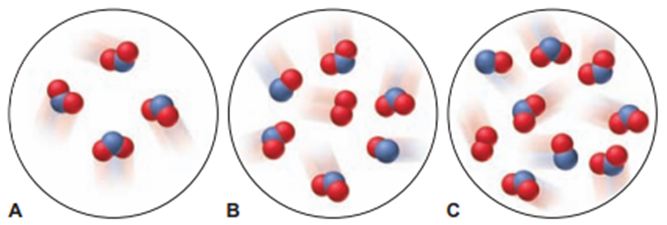
Nitrogen monoxide reacts with elemental oxygen to form nitrogen dioxide. The scene at right represents an initial mixture of reactants. If the reaction has a 66% yield, which of the scenes below (A, B, or C) best represents the final product mixture?
What do you think about this solution?
We value your feedback to improve our textbook solutions.