Chapter 3: Q3.137CP (page 138)
Elements X (green) and Y (purple) react according to the following equation: . Which molecular scene represents the product of the reaction?
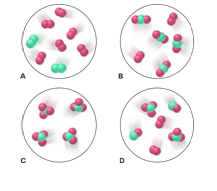
Short Answer
The molecular scene “C” represents the product of the reaction.
Learning Materials
Features
Discover
Chapter 3: Q3.137CP (page 138)
Elements X (green) and Y (purple) react according to the following equation: . Which molecular scene represents the product of the reaction?

The molecular scene “C” represents the product of the reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Seawater is approximately 4.0% by mass dissolved ions. About 85% of the mass of the dissolved ions is from NaCl.
(a) Find the mass % of NaCl in seawater.
(b) Find the mass % of Na+ions and of Cl-ions in seawater.
(c) Find the molarity of NaCl in seawater at 150C (d of seawater at 150C = 1.025 g/mL).
Hemoglobin is 6.0% heme (C34H32FeN4O4) by mass. To remove the heme, hemoglobin is treated with acetic acid and NaCl to form hemin (C34H32N4O4FeCl). At a crime scene, a blood sample contains 0.65 g of hemoglobin. (a) How many grams of heme are in the sample? (b) How many moles of heme? (c) How many grams of Fe? (d) How many grams of hemin could be formed for a forensic chemist to measure?
Are the following instructions for diluting a 10.0 M solution to a 1.00 M solution correct: “Take 100.0 mL of the 10.0 M solution and add 900.0 mL water”? Explain.
Calculate each of the following:
a). Mass % of H in ammonium bicarbonate
b). Mass % of O in sodium dihydrogen phosphate heptahydrate
Reactants D and E form product F. Write a detailed Plan to find the mass of F when 27 g of D reacts with 31 g of E.
What do you think about this solution?
We value your feedback to improve our textbook solutions.