Chapter 3: Q22P (page 131)
Oxygen is required for the metabolic combustion of foods. Calculate the number of atoms in 38.0 g of oxygen gas, the amount absorbed from the lungs at rest in about 15 min.
Short Answer
The number of atoms is
Learning Materials
Features
Discover
Chapter 3: Q22P (page 131)
Oxygen is required for the metabolic combustion of foods. Calculate the number of atoms in 38.0 g of oxygen gas, the amount absorbed from the lungs at rest in about 15 min.
The number of atoms is
All the tools & learning materials you need for study success - in one app.
Get started for free
Reactants D and E form product F. Write a detailed Plan to find the mass of F when 27 g of D reacts with 31 g of E.
The zirconium oxalate K2Zr(C2O4)3(H2C2O). H2O was synthesized by mixing 1.68 g of ZrOCl2 .8H2O with 5.20 g of H2C2O4.2H2O and an excess of aqueous KOH. After 2 months, 1.25 g of crystalline product was obtained, as well as aqueous KCl and water. Calculate the percent yield.
Question. In the process of balancing the equation
Student I writes:
Student II writes:
Student III writes:
Is the approach of Student I valid? Student II? Student III? Explain.
Write a balanced equation for the reaction depicted below:
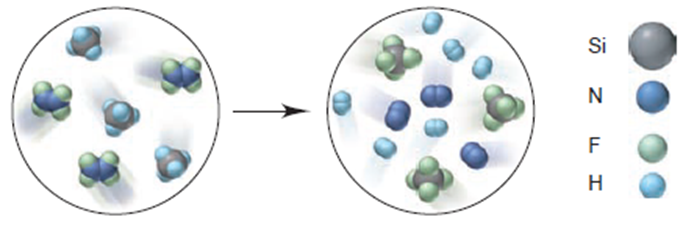
If each reactant molecule represents 1.25×10-2 mol and the reaction yield is 87%, how many grams of Si-containing product form?
Assuming that the volume are additive, what is the concentration of KBr in a solution prepared by mixing 0.200 L of 0.053 M KBr with 0.550 L of 0.078 M KBr?
What do you think about this solution?
We value your feedback to improve our textbook solutions.