Chapter 3: Q20P (page 131)
Calculate each of the following:
- Mass fraction of C in cesium acetate
- Mass fraction of O in uranyl sulfate trihydrate
Short Answer
- Mass fraction of C is 0.125
- Mass fraction of O is 0.343
Learning Materials
Features
Discover
Chapter 3: Q20P (page 131)
Calculate each of the following:
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate each of the following quantities:Number of atoms in 0.0015 mol of fluorine gas
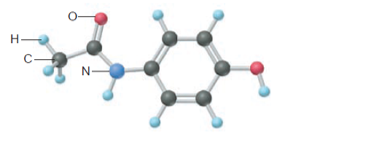
Acetaminophen (below)is a popular nonaspirin, “over-thecounter” pain reliever. What is the mass % of each element in acetaminophen?
Iron reacts slowly with oxygen and water to form a compound commonly called rust. For 45.2 kg of rust, Calculate
Potassium nitrate decomposes on heating, producing potassium oxide and gaseous nitrogen and oxygen:
To produce 56.6 kg of oxygen, how many
(a) Moles of must be heated?
(b) Grams of must be heated?
The following circles represent a chemical reaction between AB2 and B2:
(a) Write a balanced equation for the reaction. (b) What is the limiting reactant? (c) How many moles of product can be made from 3.0 mol of B2 and 5.0 mol of AB2? (d) How many moles of excess reactant remain after the reaction in part (c)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.