Chapter 14: Q14.89 (page 623)
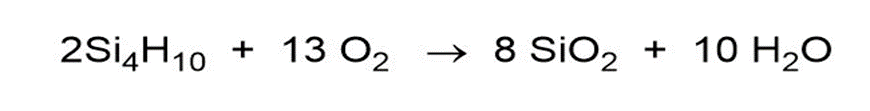
Question: One similarity between B and Si is the explosive combustion of their hydrides in air. Write balanced equations for the combustion of B2H6 and of Si4H10.
Short Answer
Answer
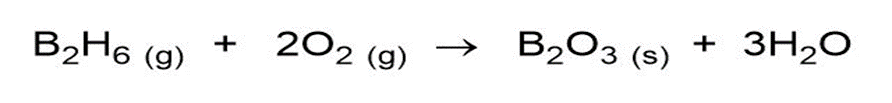
Learning Materials
Features
Discover
Chapter 14: Q14.89 (page 623)
Question: One similarity between B and Si is the explosive combustion of their hydrides in air. Write balanced equations for the combustion of B2H6 and of Si4H10.
Answer
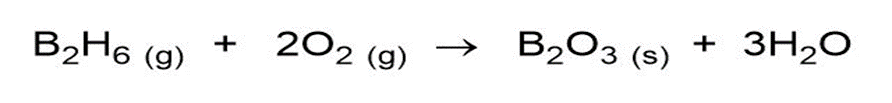

All the tools & learning materials you need for study success - in one app.
Get started for free
Which noble gas is the most abundant in the universe? In Earth’s atmosphere?
Hydrogen has only one proton, but its IE1is much greater than that of lithium, which has three protons. Explain.
(a) Why do stable xenon fluorides have an even number of atoms?
(b) Why do the ionic speciesrole="math" localid="1663319658268" and role="math" localid="1663319666688" have odd numbers of atoms?
(c) Predict the shape of.
Write balanced chemical equations for the thermal decomposition of potassium nitrate (oxygen is also formed in both the cases): (a) at low temperature to the nitrite; (b) at high temperature to the metal oxide and nitrogen.
Which Group 5A (15) elements form trihalides? Pentahalides? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.