Chapter 14: Q14.34 P (page 621)
Sketch a periodic table, and label the areas containing elements that give rise to the three types of hydrides.
Short Answer
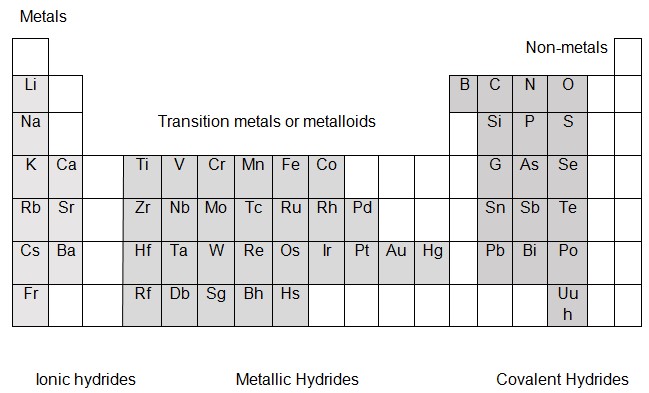
The three types of hydrides are labelled below in the periodic table:
Learning Materials
Features
Discover
Chapter 14: Q14.34 P (page 621)
Sketch a periodic table, and label the areas containing elements that give rise to the three types of hydrides.
The three types of hydrides are labelled below in the periodic table:

All the tools & learning materials you need for study success - in one app.
Get started for free
(a) How does the metallic character of an element correlate with the acidity of its oxide?
(b) What trends, if any, exist in oxide basicity across a period and down a group
Why do the noble gases have such low boiling points?
Iodine monochloride and elemental bromine have nearly the same molar mass and liquid density but very different boiling points.
(a) What molecular property is primarily responsible for this difference in boiling point? What atomic property gives rise to it? Explain.
(b) Which substance has a higher boiling point? Why?
Explain the horizontal irregularity in the size of the most common ions of Period 3 elements.
How are bond energy, bond length, and reactivity related to similar compounds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.