Chapter 24: Q24.57 P (page 1104)
What is a cation-electron pair, and how does it form?
Short Answer
Cation-electron pair is formed when a nucleus is hit with ionizing radiation.
Learning Materials
Features
Discover
Chapter 24: Q24.57 P (page 1104)
What is a cation-electron pair, and how does it form?
Cation-electron pair is formed when a nucleus is hit with ionizing radiation.
All the tools & learning materials you need for study success - in one app.
Get started for free
The isotopic mass of is 209.989669 . When this nuclide decays by electron capture, it emits2.368MeV. What is the isotopic mass of the resulting nuclide?
What are the expected advantages of fusion reactors over fission reactors?
begins a decay series that ultimately forms 206Pb . The scene below depicts the relative number of atoms (red) and atoms (green) in a mineral. If all the Pb comes from , calculate the age of the sample.
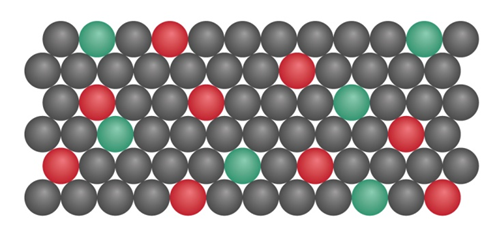
Question: Why is helium found in deposits of uranium and thorium ores? What kind of radioactive emission produces it?
Compare the s- and r-processes in stellar nucleosynthesis.
What do you think about this solution?
We value your feedback to improve our textbook solutions.