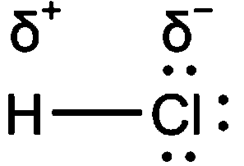The electronegativity of the atom depends upon the size, i.e., small size atoms are more electronegative and the nature of the atom. Higher the electronegativity closer the electron cloud to the nucleus. The polarity in the bonding of the molecule depends upon thedifference in the electronegativity of the atom involved.
In a partial ionic bond, there is the formation of induced cations and anions. The more negative the anion, the more polar is the bond. If the anion is more electronegative then the difference in the electronegativity increase. As the difference in electronegativity increases in the bond, the bond becomes more polar.
For example, the electronegativity difference between Sodium and Chlorine is greater than that of the difference between hydrogen and chlorine.

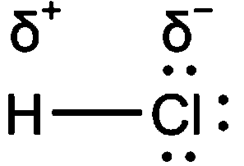
Therefore, NaCl is ionic and HCl is a polar covalent bond.