Chapter 19: Q19.67P (page 874)
Write the ion-product expressions for
Short Answer
The ion-product expression of the compounds is,
Learning Materials
Features
Discover
Chapter 19: Q19.67P (page 874)
Write the ion-product expressions for
The ion-product expression of the compounds is,
All the tools & learning materials you need for study success - in one app.
Get started for free
Why does the color change of an indicator take place over a range of about 2 pH units?
A buffer that contains base, B, and of its conjugate acid, , has a pHof . What is the pHafter mol of HCLis added to of this solution?
is purified by addingto a saturated solution of. Whenof is added to of saturated solution, what mass (g) of pure precipitates?
Explain how strong acid–strong base, weak acid–strong base, and weak base–strong acid titrations using the same concentrations differ in terms of (a) the initial pH and (b) the pH at the equivalence point. (The component in italics is in the flask.)
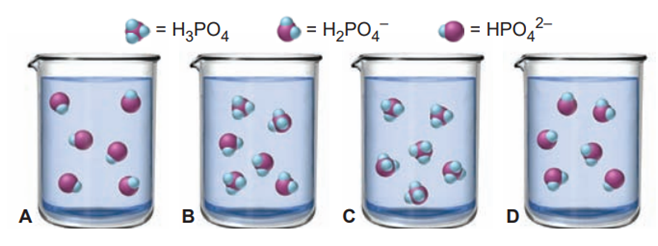
The scenes below depict the relative concentrations of , and during a titration with aqueous , but they are out of order. (Phosphate groups are purple, hydrogens are blue, and ions and water molecules are not shown.)
(a) List the scenes in the correct order.
(b) What is the pH in the correctly ordered second scene (see Appendix C)?
(c) If it requires of the solution to reach this scene, how much more is needed to reach the last scene?
What do you think about this solution?
We value your feedback to improve our textbook solutions.