Chapter 12: Q12.142CP (page 499)
KF has the same type of crystal structure as NaCl. The unit cell of KF has an edge length of 5.39 . Find the density of KF.
Short Answer
The density of KF is.
Learning Materials
Features
Discover
Chapter 12: Q12.142CP (page 499)
KF has the same type of crystal structure as NaCl. The unit cell of KF has an edge length of 5.39 . Find the density of KF.
The density of KF is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Two important characteristics used to evaluate risks of fire or explosion are a compound’s lower flammable limit (LFL) and flash point. The LFL is the minimum percentage by volume in air that is ignitable. Below that, the mixture is too “lean” to burn. The flash point is the temperature at which the air over a confined liquid becomes ignitable. n-Hexane boils at at 1 atm. At , its vapour pressure is 121 mmHg. The LFL of n-hexane is 1.1%. Calculate the flash point of n-hexane.
Bismuth is used to calibrate instruments employed in high-pressure studies because it has several well-characterized crystalline phases. Its phase diagram (right) shows the liquid phase and five solid phases that are stable above 1 katm (1000 atm) and up to .
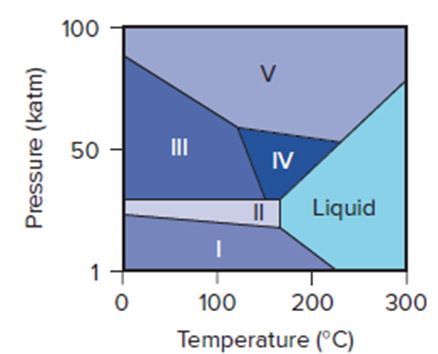
(a) Which solid phases are stable at?
(b) Which phase is stable at 50 katm and?
(c) As the pressure is reduced from 100 to 1 katm at, what phase transitions does bismuth undergo?
(d) What phases are present at each of the triple points?
Why are units of energy per area used for surface tension values?
Zinc selenide (ZnSe) crystallizes in the zinc blende structure (see Figure) and has a density of 5.42 g/cm3.
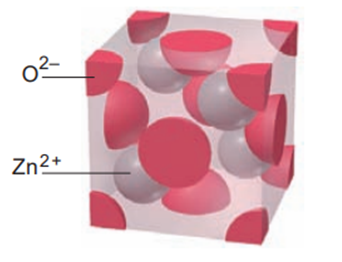
(a) How many Zn and Se ions are in each unit cell?
(b) What is the mass of a unit cell?
(c) What is the volume of a unit cell?
(d) What is the edge length of a unit cell?
The phase diagram for substance A has a solid-liquid line with a positive slope, and that for substance B has a solid-liquid line with a negative slope. What macroscopic property can distinguish A from B?
What do you think about this solution?
We value your feedback to improve our textbook solutions.