Chapter 12: 12.91P (page 497)
Calcium sulfide adopts the sodium chloride crystal structure (Figure). How manyions are in the CaS unit cell?
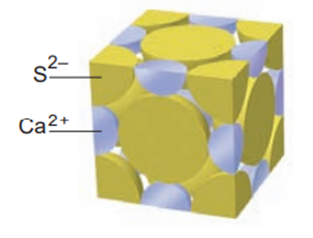
Short Answer
Four ions are present in the CaS unit cell
Learning Materials
Features
Discover
Chapter 12: 12.91P (page 497)
Calcium sulfide adopts the sodium chloride crystal structure (Figure). How manyions are in the CaS unit cell?

Four ions are present in the CaS unit cell
All the tools & learning materials you need for study success - in one app.
Get started for free
In the photoelectric effect, the work function (Φ) is the minimum energy a photon must have to remove an electron from a metal surface (see Problem 7.70). For a given metal, Φ depends on how and where the photon strikes the crystal. Copper adopts the face-centered cubic structure. If the photon strikes perpendicular to the unit cell’s face, Φ is 4.59 eV; if perpendicular to a plane tangent to an edge, Φ is 4.48 eV; and if perpendicular to a plane tangent to a corner, Φ is 4.94 eV (1 eV = ). Find the wavelength (in nm) of the lowest energy photon that can remove an electron from copper, and state how it strikes the unit cell.
Which has the greater polarizability? Explain.
(a) or Ca (b) or (c) or
Rank the compounds in Problem 12.61 in order of decreasing viscosity at a given temperature; explain your ranking.
Of the five major types of crystalline solid, which does each of the following forms, and why: (a) SiC; (b) Na2SO4; (c) SF6; (d) cholesterol (C27H45OH) ; (e) KCl; (f) BN?
Which has the greater polarizability? Explain.
(a) or (b) or (c) or
What do you think about this solution?
We value your feedback to improve our textbook solutions.