Chapter 12: 12.87P (page 496)
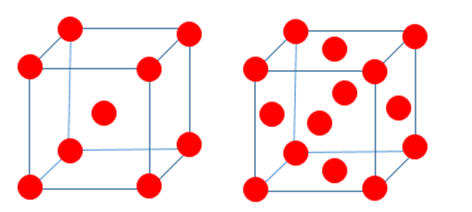
As molten iron cools to 1674 K, it adopts one type of cubic unit cell; then, as the temperature drops below 1181 K, it changes to another, as depicted below:
- What is the change in unit cell?
- Which crystal structure has the greater packing efficiency?
Short Answer
- When molten iron cools to 1674 K, it has face-centered cubic unit cell; whenever the temperature comes below 1181 K, it changes to body-centered cubic unit cell.
- Face-centered crystal structure has greater packing efficiency.