Chapter 12: 12.15_P (page 494)
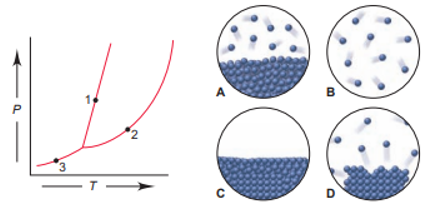
Match each numbered point in the phase diagram for compound Q with the correct molecular depiction below:
Short Answer
The match for the numbers are:
1= D
2= A
3= C
Learning Materials
Features
Discover
Chapter 12: 12.15_P (page 494)
Match each numbered point in the phase diagram for compound Q with the correct molecular depiction below:

The match for the numbers are:
1= D
2= A
3= C
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Why are gases more easily compressed than liquids?
(b) Why do liquids have a greater ability to flow than solids?
Does the strength of the intermolecular forces in a liquid change as the liquid is heated? Explain. Why does liquid viscosity decrease with rising temperature?
What is the of a liquid that has a vapour pressure of 621 torr at and a boiling point of at 1 atm?
A liquid is in equilibrium with its vapour in a closed vessel at a fixed temperature. The vessel is connected by a stopcock to an evacuated vessel. When the stopcock is opened, will the final pressure of the vapour be different from the initial value if
(a) Some liquid remains
(b) All the liquid is first removed?
Why does the antifreeze ingredient ethylene glycol ( ) have a boiling point of 197.6°C, whereas propanol ( ), a compound with a similar molar mass, has a boiling point of only 97.4°C?
What do you think about this solution?
We value your feedback to improve our textbook solutions.