Chapter 12: 12.102P (page 497)
When tin is added to copper, the resulting alloy (bronze) is much harder than copper. Explain.
Short Answer
An alloy is formed when two or more metals are mixed in a certain proportion.
Learning Materials
Features
Discover
Chapter 12: 12.102P (page 497)
When tin is added to copper, the resulting alloy (bronze) is much harder than copper. Explain.
An alloy is formed when two or more metals are mixed in a certain proportion.
All the tools & learning materials you need for study success - in one app.
Get started for free
Butane is a common fuel used in cigarette lighters and camping stoves. Normally supplied in metal containers under pressure, the fuel exists as a mixture of liquid and gas, so high temperatures may cause the container to explode. At , the vapour pressure of butane is 2.3 atm. What is the pressure in the container at vap ( =24.3 kJ/mol)?
Does the strength of the intermolecular forces in a liquid change as the liquid is heated? Explain. Why does liquid viscosity decrease with rising temperature?
Explain the effect of strong intermolecular forces on each of these parameters:
(a) Critical temperature
(b) Boiling point
(c) Vapor pressure
(d) Heat of vaporization
Which member of each pair of compounds forms intermolecular H bonds? Draw the H-bonded structures in each case:
(a) or (b) data-custom-editor="chemistry" or
Bismuth is used to calibrate instruments employed in high-pressure studies because it has several well-characterized crystalline phases. Its phase diagram (right) shows the liquid phase and five solid phases that are stable above 1 katm (1000 atm) and up to .
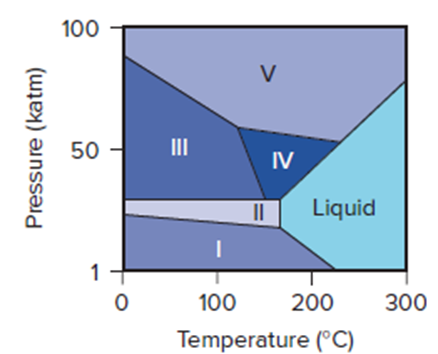
(a) Which solid phases are stable at?
(b) Which phase is stable at 50 katm and?
(c) As the pressure is reduced from 100 to 1 katm at, what phase transitions does bismuth undergo?
(d) What phases are present at each of the triple points?
What do you think about this solution?
We value your feedback to improve our textbook solutions.