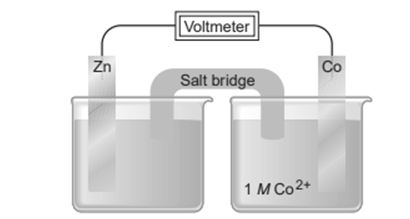
Chapter 21: Q21.141 CP (page 978)
You are given the following three half-reactions:
(a). Use values for ( 1 ) and ( 2 ) to find for ( 3 ).
(b) Calculate for ( 1) and ( 2) from their values.
(c) Calculate for (3) from (1) and ( 2 ).
(d) Calculate for ( 3) from its .
(e) What is the relationship between the values for (1) and (2) and thevalue for (3 )?
Short Answer
- The values of are obtained as: .
- The value of are obtained as: .
- The value of is obtained as: .
- The value of from for the third reaction is obtained as: .
- The Gibbs Free Energy calculation may be utilised to derive the half-cell potential for (3 ) using the half-cell potentials for ( 1 ) and ( 2 ).