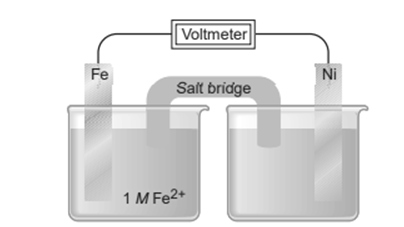
Chapter 21: Q21.137 CP (page 978)
In an electric power plant, personnel monitor the content of boiler feed water to prevent corrosion of the boiler tubes. Why does Fe corrode faster in steam and hot water than in cold water?
Short Answer
The activation energy of the reactants is easier to obtain at higher temperatures, hence Fe corrodes quicker in high-temperature fluids or steam.