Chapter 9: Q. 9.54 (page 306)
A patient received in . How many milliliters of a (saline) solution were delivered?
Short Answer
of a saline solution were delivered.
Learning Materials
Features
Discover
Chapter 9: Q. 9.54 (page 306)
A patient received in . How many milliliters of a (saline) solution were delivered?
of a saline solution were delivered.
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the difference between a methanol solution and a methanol solution?
What is the difference between a glucose solution and a glucose solution?
Calculate the mass percent of a solution containing localid="1654600997108"
In the following diagram, blue spheres represent the element and yellow spheres represent the element .
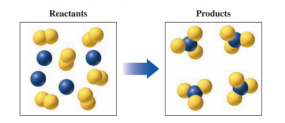
a. Write the formulas for each of the reactants and products.
b. Write a balanced chemical equation for the reaction.
c. Indicate the type of reaction as combination, decomposition, single replacement, double replacement, or combustion.
Predict whether each of the following ionic compounds is soluble in water:
a.
b.
c.
d.
e.
What do you think about this solution?
We value your feedback to improve our textbook solutions.