Chapter 5: Q.5.79 (page 165)
Write the balanced nuclear equation for each of the following radioactive emissions:
a. an alpha particle from Hg-180
b. a beta particle from Au-198
c. a positron from Rb- 82
Short Answer
Part (a)
Part (b)
Part (c)
Learning Materials
Features
Discover
Chapter 5: Q.5.79 (page 165)
Write the balanced nuclear equation for each of the following radioactive emissions:
a. an alpha particle from Hg-180
b. a beta particle from Au-198
c. a positron from Rb- 82
Part (a)
Part (b)
Part (c)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the nucleus of the isotope that is bombarded in the following:
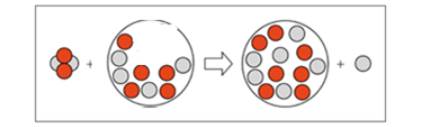
Complete the following fission reaction.
Xenon-133 is used to test lung function; it decays by emitting a beta particle.
a. Write an equation for the beta decay of Xe-133.
b. If the half-life of Xe-133 is 5.2h, how much of a 20 .-mCi sample is still active after 15.6 h?
A sample of sodium-24 with an activity of 12 mCi was used to study the rate of blood flow in the circulatory system. If sodium-24 has a half-life of 15h, what is the activity after each of the following intervals?
a. one half-life
b. 30h
c. three half-lives
d. 2.5 days
Calcium-, used to evaluate bone metabolism, has a half-life of days.
a. Write the balanced nuclear equation for the beta decay of calcium-.
b. How many milligrams of a sample of calcium- remain after days?
c. How many days have passed ifof calcium- decayed to of calcium-?
What do you think about this solution?
We value your feedback to improve our textbook solutions.