Chapter 5: Q.5.54 (page 164)
Complete the following bombardment reaction by drawing the nucleus of the new isotope that is produced in the following:
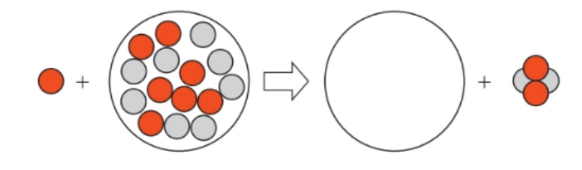
Short Answer
The reaction is as follows,
Learning Materials
Features
Discover
Chapter 5: Q.5.54 (page 164)
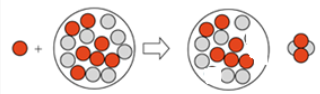
Complete the following bombardment reaction by drawing the nucleus of the new isotope that is produced in the following:

The reaction is as follows,

All the tools & learning materials you need for study success - in one app.
Get started for free
Match the type of radiation(to ) with each of the following statement:
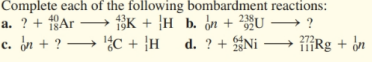
Element 114 was recently named flerovium, symbol F. The reaction for its synthesis involves bombarding Pu-244 with Ca-48. Write the balanced nuclear equation for the synthesis of flerovium.
Write a balanced equation for the beta decay of each of the following radioactive isotopes:
(a) silicon-
(b) cobalt-
(c)data-custom-editor="chemistry"
(d)data-custom-editor="chemistry"
The radioisotope sodium-24 is used to determine the levels of electrolytes in the body. A 16sample of sodium-24 decays to 2.0 in 45 h. What is the half-life, in hours, of sodium-24?
What do you think about this solution?
We value your feedback to improve our textbook solutions.