Chapter 5: Q. 5.76 (page 165)
If the amount of radioactive iodine-, used to treat thyroid cancer, in a sample decreases from my to in , what is the half-life of iodinc-? (5.4)
Short Answer
The iodin half-life-is
Learning Materials
Features
Discover
Chapter 5: Q. 5.76 (page 165)
If the amount of radioactive iodine-, used to treat thyroid cancer, in a sample decreases from my to in , what is the half-life of iodinc-? (5.4)
The iodin half-life-is
All the tools & learning materials you need for study success - in one app.
Get started for free
a. What are the products in the fission of uranium-that make possible a nuclear chain reaction?
b. What is the purpose of placing control rods among uranium samples in a nuclear reactor?
Write the balanced nuclear equation for each of the following:
a. When two oxygen-atoms collide, one of the products is an alpha particle.
b. When californium- is bombarded by oxygen- , a new element, seaborgium-, and four neutrons are produced.
c. Radon- undergoes alpha decay.
d. An atom of strontium- emits a positron.
Draw the nucleus of the isotope that is bombarded in the following:
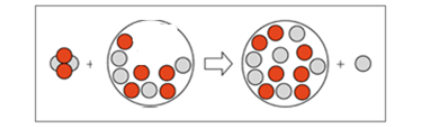
Complete each of the following nuclear equations:
b.
c.
d.
A nuclear technician was accidentally exposed to potassium-42 while doing brain scans for possible tumours. The error was not discovered until 36 h later when the activity of the potassium-42 sample was 2.0 Ci. If potassium- 42 has a half-life of 12 h, what was the activity of the sample at the time the technician was exposed?
What do you think about this solution?
We value your feedback to improve our textbook solutions.