Chapter 5: Q. 5.7 (page 140)
Write the atomic symbol for each of the following isotopes used in nuclear medicine:
a.
b.
c.
d.
Short Answer
Part(a) Atomic symbol is
Part(b) Atomic symbol is
Part(c) Atomic symbol is
Part(d) Atomic symbol is
Learning Materials
Features
Discover
Chapter 5: Q. 5.7 (page 140)
Write the atomic symbol for each of the following isotopes used in nuclear medicine:
a.
b.
c.
d.
Part(a) Atomic symbol is
Part(b) Atomic symbol is
Part(c) Atomic symbol is
Part(d) Atomic symbol is
All the tools & learning materials you need for study success - in one app.
Get started for free
Match each property ( to ) with its unit of measurement.
1. activity
2. absorbed done
3. biological damage
a. rad
b. mrem
c. mCi
d. Gy
All the elements beyond uranium, the trans uranium elements, have been prepared by bombardment and are not naturally occurring elements. The first trans uranium element neptunium, , was prepared by bombarding with neutrons to form a neptunium atom and a beta particle. Complete the following equation:
data-custom-editor="chemistry"
Calcium-, used to evaluate bone metabolism, has a half-life of days.
a. Write the balanced nuclear equation for the beta decay of calcium-.
b. How many milligrams of a sample of calcium- remain after days?
c. How many days have passed ifof calcium- decayed to of calcium-?
Xenon-133 is used to test lung function; it decays by emitting a beta particle.
a. Write an equation for the beta decay of Xe-133.
b. If the half-life of Xe-133 is 5.2h, how much of a 20 .-mCi sample is still active after 15.6 h?
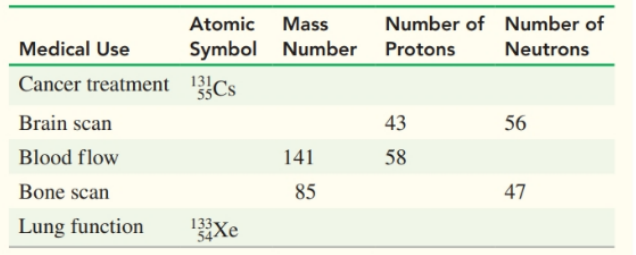
Supply the missing information in the following table:
What do you think about this solution?
We value your feedback to improve our textbook solutions.