Chapter 3: Q.3.83 (page 93)
What is in degrees Celsius and in kelvins?
Learning Materials
Features
Discover
Chapter 3: Q.3.83 (page 93)
What is in degrees Celsius and in kelvins?
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate the heat change at for each of the following and indicate whether heat was absorbed/released:
a. calories to vaporize g of water
b. joules to vaporize g of water
c. kilocalories to condense kg of steam
d. kilojoules to condense g of steam
Identify each of the following changes of stane as melting. freezing, sublimation, or deposition:
a. The solid structure of a substance breaks down as liquid forms.
b. Coffee is freeze-dried.
c. Water on the street turns to ice during a cold wintry night.
d. Ice crystals form on a package of frozen corm.
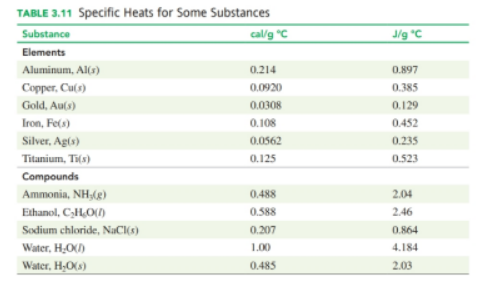
Use the heat equation to calculate the energy for each of the following (see Table):
a. calories lost whenlocalid="1653051789792" a of waler cools from localid="1651743554041" to localid="1651743558077"
h. joules to heatlocalid="1651743562257" of water from localid="1651743566051" to localid="1651743570658"
C. kilocalories to heat localid="1651743574953" of water from localid="1651743579245" to localid="1651743583100"
d. kilojoules to heat localid="1651743586855" of gold fromlocalid="1651743591821" to localid="1651743595595"
Classify each of the following pure substances as an element or a compound:
a. silicon () chip
b. hydrogen peroxide localid="1651737397761"
c. oxygen gas localid="1651737412373" role="math"
d. rust localid="1651737418279"
e. methane localid="1651737422197" in natural gas
A patient receives of intravenous () glucose solution. If of the solution contains of glucose (carbohy. drate), how many kilocalories did the patient obtain from the glucose solution?
What do you think about this solution?
We value your feedback to improve our textbook solutions.