Chapter 15: Q. 15.21 (page 521)
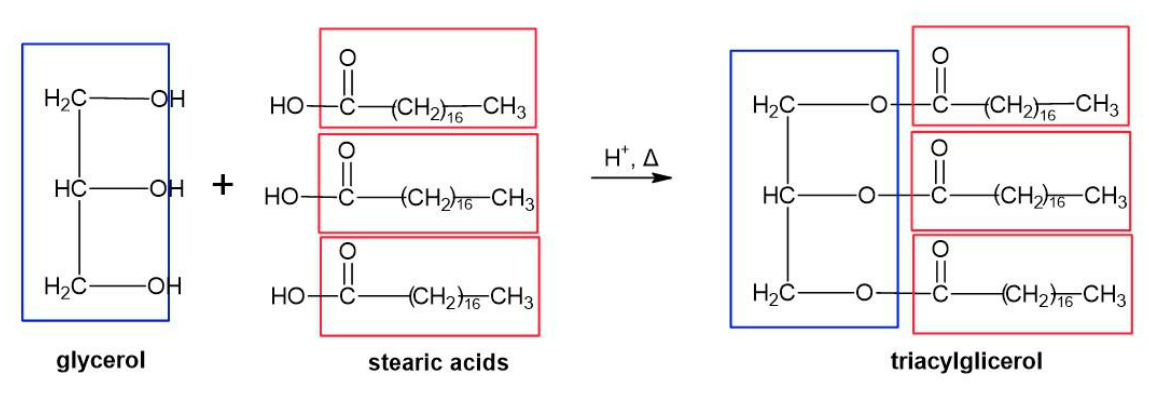
Draw the condensed structural formula for a triacylglycerol that contains stearic acid and glycerol.
Short Answer
Learning Materials
Features
Discover
Chapter 15: Q. 15.21 (page 521)
Draw the condensed structural formula for a triacylglycerol that contains stearic acid and glycerol.

All the tools & learning materials you need for study success - in one app.
Get started for free
The total kilocalories and grams of fat for some typical meals at fast-food restaurants are listed here. Calculate the number of kilocalories and the percentage of total kilocalories from fat . Round answers to the tens place.
a. a chicken dinner, of fat
b. a quarter-pound cheeseburger, role="math" localid="1653102911440" of fat
c. pepperoni pizza (three slices), of fat
What are some sources of omega- and omega- -fatty acids (see Chemistry Link to Health "Omega-Fatty Acids in Fish Oils")?
How does the double bond influence the dispersion forces that can form between the hydrocarbon chains of fatty acids?
Among the ingredients in lipstick are beeswax, carnauba wax, hydrogenated vegetable oils, and glyceryl tricaprate (tricaprin).
a. What types of lipids are these?
b. Draw the condensed structural formula for glyceryl tricaprate (tricaprin). Capric acid is a saturated carbon fatty acid.
Olive oil is monounsaturated, whereas hutter fot is caturated. Why does olive oil have a lower meltiag point than butser far?
What do you think about this solution?
We value your feedback to improve our textbook solutions.