Chapter 6: Q. 6.147 (page 210)
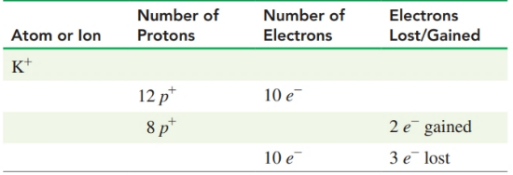
Complete the following table for atoms or ions:
Short Answer
| Atom or Ion | Number of protons | Number of Electron | Electrons loss/ Gained |
| p | lost | ||
| p | lost | ||
| p | gained | ||
| p | lost |
Learning Materials
Features
Discover
Chapter 6: Q. 6.147 (page 210)
Complete the following table for atoms or ions:

| Atom or Ion | Number of protons | Number of Electron | Electrons loss/ Gained |
| p | lost | ||
| p | lost | ||
| p | gained | ||
| p | lost |
All the tools & learning materials you need for study success - in one app.
Get started for free
Write the formula for each of the following molecular compounds:
a. silicon dioxide
b. carbon tetrabromide
c. diphosphorus tetraiodide
d. dinitrogen trioxide
Which of the following pairs of elements are likely lo form an ionic compound?
a. helium and oxygen
b. magnesium and chlorine
c. chlorine and bromine
d. potassium and sulfur
e. sodium and potassium
f. nitrogen and iodine
Name each of the following ionic compounds:
a. , antidepressant
b. , Epsom salts
c. , disinfectant
d. , laxative
e. , component of antacids
Identify the strongest intermolecular forces between the particles of each of the following:
a.
b.
c.
d.
Which electronegativity difference (a, b, or c) would you expect for a nonpolar covalent bond?
(a) from to
(b) from to
(c) from to
What do you think about this solution?
We value your feedback to improve our textbook solutions.