Chapter 6: Q. 6.134 (page 210)
Show the dipole arrow for each of the following bonds:
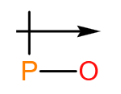
a.
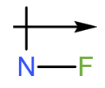
b.
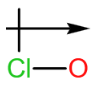
c.
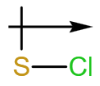
d.
e.
Short Answer
a. The dipole arrow of is .
.
b. The dipole arrow of is  .
.
c. The dipole arrow of is .
.
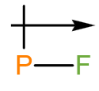
d. The dipole arrow of is 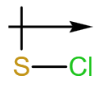
e. The dipole arrow of is .
.