Chapter 6: Q. 6.127 (page 210)
Draw the Lewis structure for each of the following:
a.
b.(is the central atom)
c.
Short Answer
(a) The Lewis structure of is
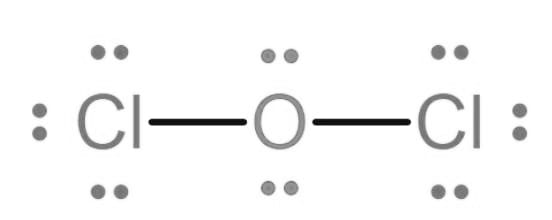
(b) The Lewis structure of is
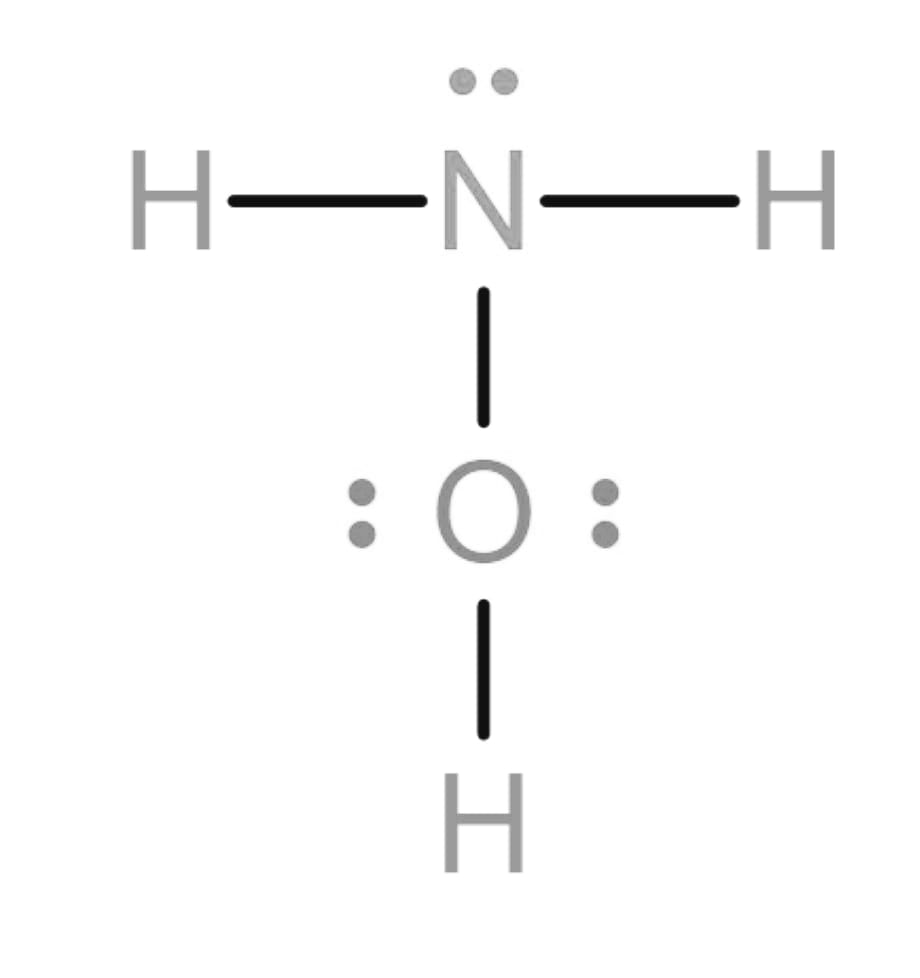
(c) The Lewis structure of is
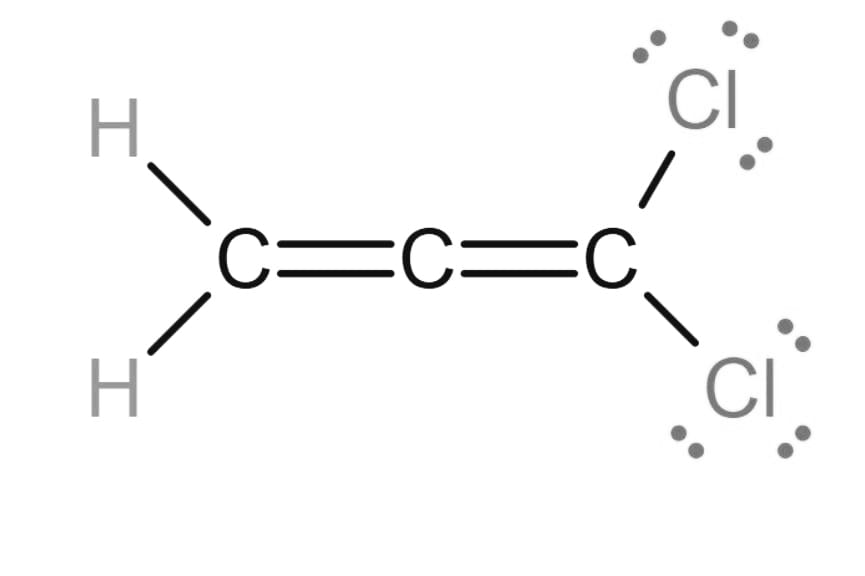
Learning Materials
Features
Discover
Chapter 6: Q. 6.127 (page 210)
Draw the Lewis structure for each of the following:
a.
b.(is the central atom)
c.
(a) The Lewis structure of is

(b) The Lewis structure of is

(c) The Lewis structure of is

All the tools & learning materials you need for study success - in one app.
Get started for free
Identify each of the following atoms or ions:
Name each of the following ionic compounds:
a. , antidepressant
b. , Epsom salts
c. , disinfectant
d. , laxative
e. , component of antacids
Complete each of the following statements for a molecule of :
a. There are electron groups around the centrallocalid="1652416979713" atom.
b. The electron-group geometry is .
c. The number of atoms attached to the central localid="1652416983498" atom is
d. The shape of the molecule is .
State the number of valence electrons, bonding pairs, and lone pairs in each of the following Lewis structures:
Write the name for each of the following ionic compounds:
a.
b.
c.
d.
e.
f.
What do you think about this solution?
We value your feedback to improve our textbook solutions.