Chapter 11: Q. 11.58 (page 394)
Draw the cis and trans forms for each of the following:
a) Butene
b) hexene
Short Answer
The cis and trans forms are:
a)
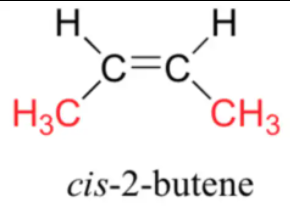
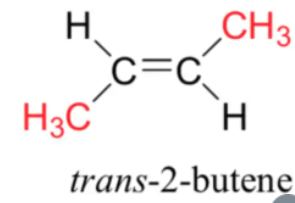
b)
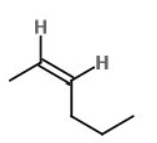

Learning Materials
Features
Discover
Chapter 11: Q. 11.58 (page 394)
Draw the cis and trans forms for each of the following:
a) Butene
b) hexene
The cis and trans forms are:
a)


b)


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structural formula for the product in each of the following reactions:
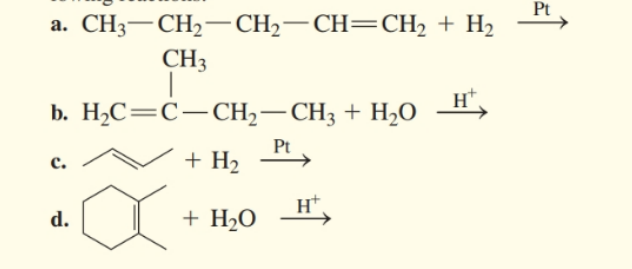
Match the following physical and chemical properties with pottasium chloride, KCL, used in salt substances, or butane used in lighters.

a) melts at
b) burn vigorously in air
c) melts at
d) contains iconic bonds
e) is a gas at room temperature.
Consider the compound propane.
a. Draw the condensed structural formula for propane.
b. Write the balanced chemical equation for the complete combustion of propane.
c. How many grams of are needed to react with of propane gas at STP?
d. How many grams of would be produced from the reaction in part c?
Draw the condensed structural formula for alkanes or the line angle formula for cycloalkanes for each of the following:
a.
b.
c.
d.
Draw the condensed structural or line-angle formula if cyclic, for each of the following:
a.
b.
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.