Chapter 8: Q. 8.68 (page 282)
Indicate if pressure increase decreases, or stays the same in each of the following:

Short Answer
a. The pressure will increase.
b. The pressure will increase.
c. The pressure will increase.
Learning Materials
Features
Discover
Chapter 8: Q. 8.68 (page 282)
Indicate if pressure increase decreases, or stays the same in each of the following:

a. The pressure will increase.
b. The pressure will increase.
c. The pressure will increase.
All the tools & learning materials you need for study success - in one app.
Get started for free
A typical air sample in the lungs contains oxygen at , nitrogen at ,n carbon dioxide at , and water vapor at . Why are these pressures called partial pressures?
Indicate which diagram represents the volume of the gas sample in a flexible container when each of the following changes takes place:
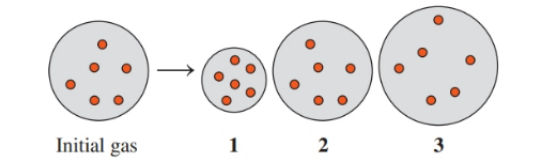
a. Temperature increases if the pressure does not change.
b. Temperature decreases if the pressure does not change.
c. Atmospheric pressure decreases if the temperature does not change.
d. Doubling the atmospheric pressure and doubling the Kelvin temperature.
Sometimes when you blow up a balloon and release it, it flies around the room. What is happening to the air in the balloon and its volume?
An air bubble has a volume of at What is the final volume, in liters, of the gas when the temperature changes to each of the following, if and do not change?
a.
b.
c.
d.
A gas sample has a volume of with an unknown temperature. The same gas has a volume of when the temperature is , with no change in the pressure or amount of gas. What was the initial temperature, in degrees Celsius, of the gas?
What do you think about this solution?
We value your feedback to improve our textbook solutions.